Salt content (% Cl) was
determined volumetrically by Knudsen-type titration (Strickland and Parsons,
1972) using AgNO3 solution with K2CrO4 color
indicator. The 1![]() relative standard deviation was 3%.
relative standard deviation was 3%.
Concentrations of V, Cr,
Mn, Co, Ni, Cu, Zn, As, Rb, Sr, Y, Zr, Nb, Ba, Pb, and Ti were determined using
a Phillips PW2400 X-ray spectrometer equipped with a Rh tube powered at 3 kW
following the method of Calvert et al. (1985). Precisions (1![]() relative standard deviations) were 30%-50% for Co, As, and Pb (all of which had
low concentrations), 15% for Ni, 7% for Nb, 6% for Ba, and less than 5% for the
other elements.
relative standard deviations) were 30%-50% for Co, As, and Pb (all of which had
low concentrations), 15% for Ni, 7% for Nb, 6% for Ba, and less than 5% for the
other elements.
Contents of Re, Mo, and U
were analyzed by isotope dilution flow-injection-analysis ICP-MS on a Plasmaquad
II+ in the peak-jumping mode following the method of Crusius et al. (1996).
Samples were analyzed for Re after complete microwave digestion in concentrated
HCl, HNO3, and HF, and anion exchange preconcentration using Dowex AG
1X-8 (100-200 mesh). Subsamples for Mo and U analysis were collected from a
weighed fraction of the Re digest for measurement. Precisions (1![]() relative standard deviation) are ~2% for Re and 5% for Mo and U.
relative standard deviation) are ~2% for Re and 5% for Mo and U.
Organic (Corg) and carbonate carbon (Ccarb) contents (analyzed by a LECO WR-12 carbon determinator), pyrite abundance (determined semiquantitatively by X-ray diffraction analysis), and volume percentages (vol%) of the silt-size component (determined by laser diffraction-scattering method) were determined by Tada et al. (Chap. 25, this volume). Depth profiles for these parameters are compared here with those for minor and trace element concentrations to examine relationships among the variations in minor and trace metals and those in biogenic, diagenetic, and detrital materials.
When reporting elemental
concentrations in marine sediments, salt interferes both as a direct diluent and
as a contributor of specific elements to the sediments. To correct for this, two
assumptions were made: first, that interstitial water salinity was 35![]() ,
and second, that the chlorine in the sediment is present entirely as salt and
not within any other components. Factors for salt correction were calculated
using the composition of seawater tabulated by Horne (1969). The correction for
Sr was applied first, using the following equation:
,
and second, that the chlorine in the sediment is present entirely as salt and
not within any other components. Factors for salt correction were calculated
using the composition of seawater tabulated by Horne (1969). The correction for
Sr was applied first, using the following equation:
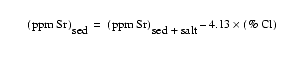 .
.
Specific salt corrections for other elements were not necessary because of their low concentrations in sea salt. All elemental concentrations were subsequently corrected for bulk dilution by salt using the equation
 .
.