RESULTS
PACMANUS comprises several discrete vent site fields (all on the order of 100–200 m in diameter) over a 3-km section of the neovolcanic Pual Ridge. Fluid temperatures measured at the orifices of black or gray smokers and sulfide chimneys venting clear fluid range between 220°C and 276°C, and end-member vent fluids are acidic (pH 2.5–3.5), show high K/Ca ratios, and are high in Mn, Fe, and Pb relative to mid-ocean ridge fluids (Gamo et al., 1996; Shitashima et al., 1997; Douville, 1999). The fluids also exhibit variable salinities, which may imply subsurface phase separation, suggesting that hydrothermal temperatures exceed 350°C below the chimney fields. Douville et al. (1999) ascribe unusually high fluorine contents in the fluids to magmatic sources. Similarly, Ishibashi et al. (1996) explain end-member gas compositions of 20–40 mM CO2, 20–40 µM CH4, and R/RA(He) = 7.4 by significant magmatic input into the hydrothermal fluids.
Drilling was attempted at the Satanic Mills site (3°43.63´S, 151°40.40´E; 1708–1720 m water depth), the Roman Ruins site (3°43.27´S, 151°40.50´E; 1693–1700 m water depth), and in an area of diffuse venting, the Snowcap site (3°43.67´S, 151°40.25´E; 1654–1670 m water depth) (Binns, Barriga, Miller, et al., 2002). The Snowcap area is a site of extensive low-temperature (6°C) diffuse venting across a 10- to 15-m-high knoll with more intense activity at the edges. Dredge and submersible sampling recovered completely hydrothermally altered dacites dominated by advanced argillic alteration to cristobalite, with lesser natroalunite, diaspore, and illite-montmorillonite (Yeats et al., 2000).
We reported data for anhydrite from Snowcap (Site 1188) and Roman Ruins (Site 1189). Two holes were drilled at each site with maximum crustal penetrations of 375 meters below sea floor (mbsf) at Site 1188 and 200 mbsf at Site 1189. Rocks encountered in all drill holes are dacitic in composition and include massive to vesicular lava flows, autoclastic breccias, and volcaniclastic sediments. Except for fresh lava flows near the seafloor, all rocks are highly to completely altered to silica (cristobalite and quartz), clay (chlorite, illite, pyrophyllite, smectite, and mixed-layer phases), and anhydrite. X-ray diffraction (XRD) results indicate that the clay composition is variable with alternating illite- and chlorite-dominated assemblages in the shallow crust at PACMANUS. Thermal gradients at Site 1189 appear to be higher than those at Site 1188, as indicated by the shallower depth at which the transition from cristobalite to quartz occurs (30 vs. 120 mbsf) (Binns, Barriga, Miller, et al., 2002).
The common occurrence of anhydrite as vein and vesicle fill, but also replacive and as breccia cement, is remarkable. Anhydrite is typically associated with pyrite and, less frequently, with quartz. It appears to be generally late in the sequence of hydrothermal alteration stages, which include early pervasive green clay-quartz alteration, localized quartz-white clay-anhydrite alteration, silicification, and late anhydrite-pyrite veining. Downhole profiles of mineral abundance suggest that the extent of silicification increases and the anhydrite contents decrease downsection in all drill holes (Binns, Barriga, Miller, et al., 2002). Moreover, relict igneous plagioclase (labradorite) is generally more abundant deeper in the sections, suggesting less intense alteration or conditions of fluid-rock reactions under which labradorite is stable.
Data are reported in Table T1 and plotted vs. depth in Figures F1, F2 and F3. This data report presents all anhydrite chemical data collected to this date. Subsets of this data collection were presented and discussed previously (Sr and S isotope compositions in Roberts et al. [2003]; REE data for Site 1188 in Bach et al. [2003]).
Figure F1 shows the downhole variability of Sr isotope compositions that vary between seawater (0.70918) (Palmer and Edmond, 1989) and an assumed hydrothermal end-member composition of 0.7050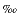 (see discussions in Roberts et al., 2003, and Bach et al., 2003). At seawater salinity, anhydrite precipitates above 140°–150°C when seawater is being heated or seawater (28 mmol/kg sulfate) mixes with nominally sulfate-free hydrothermal fluids. Sr concentrations in the PACMANUS black smoker fluids are slightly higher than those in seawater (Douville, 1999). A simple Sr mass balance provides the proportions of seawater in the fluids from which anhydrite was precipitated:
(see discussions in Roberts et al., 2003, and Bach et al., 2003). At seawater salinity, anhydrite precipitates above 140°–150°C when seawater is being heated or seawater (28 mmol/kg sulfate) mixes with nominally sulfate-free hydrothermal fluids. Sr concentrations in the PACMANUS black smoker fluids are slightly higher than those in seawater (Douville, 1999). A simple Sr mass balance provides the proportions of seawater in the fluids from which anhydrite was precipitated:
-
%seawater = (RHF x CHF – RAnhy x CHF)/
-
[RHF x CHF – RSW x CSW – RAnhy x (CHF – CSW)] x 100,
where R = 87Sr/86Sr ratio and C = Sr concentration of seawater (SW), anhydrite (Anhy), and the hydrothermal fluid end-member (HF), respectively (Table T1).
The most radiogenic values (corresponding to 84%–89% seawater) were observed in the shallowest anhydrite veins cored in Hole 1188A. As discussed in Roberts et al. (2003) and Vanko et al. (2004), the formation of these veins requires some conductive heating of seawater for anhydrite saturation to be achieved in a mix that has >84% seawater. The average percent seawater for the PACMANUS anhydrites is 40% ± 17% (1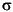 ; n = 105). By comparison, anhydrites recovered from drill core from the Trans-Atlantic Geotraverse (TAG) hydrothermal system at the Mid-Atlantic Ridge 26°N (Mills et al., 1998; Teagle et al., 1998a) precipitated from fluids with an average of 68% ± 12% seawater (1
; n = 105). By comparison, anhydrites recovered from drill core from the Trans-Atlantic Geotraverse (TAG) hydrothermal system at the Mid-Atlantic Ridge 26°N (Mills et al., 1998; Teagle et al., 1998a) precipitated from fluids with an average of 68% ± 12% seawater (1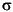 ; n = 42). Anhydrite from Site 1189 yields an average of 47% ± 16% seawater, which is somewhat higher than the average for anhydrite from Site 1188 (35% ± 17%; 33% ± 13% if the shallowest three samples are excepted).
; n = 42). Anhydrite from Site 1189 yields an average of 47% ± 16% seawater, which is somewhat higher than the average for anhydrite from Site 1188 (35% ± 17%; 33% ± 13% if the shallowest three samples are excepted).
The sulfur isotopic composition of anhydrite from Site 1188 is mostly enriched in 32S relative to seawater ( 34S = 21
34S = 21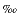 ) (Rees et al., 1978). This could be due either to oxidation of sulfide or addition of sulfate derived from disproportionation of magmatic SO2 (e.g., Gamo et al., 1997).
) (Rees et al., 1978). This could be due either to oxidation of sulfide or addition of sulfate derived from disproportionation of magmatic SO2 (e.g., Gamo et al., 1997).  34S numbers of anhydrites from Hole 1189B and the majority of samples from Hole 1189A range between 20
34S numbers of anhydrites from Hole 1189B and the majority of samples from Hole 1189A range between 20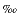 and 23
and 23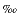 , similar to anhydrite from the TAG hydrothermal field (Chiba et al., 1998). As pointed out by Roberts et al. (2003), the
, similar to anhydrite from the TAG hydrothermal field (Chiba et al., 1998). As pointed out by Roberts et al. (2003), the  34S values of anhydrite from Site 1188 tend to be systematically lower by 1
34S values of anhydrite from Site 1188 tend to be systematically lower by 1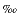 –2
–2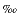 , which may relate to systematic differences in hydrothermal processes at these two sites.
, which may relate to systematic differences in hydrothermal processes at these two sites.
Sr concentrations of anhydrite are plotted vs. subbasement depth in Figure F2. Although there is some overlap, anhydrites from Site 1188 tend to have higher Sr concentrations than anhydrites from Site 1189. We used the Sr concentrations in combination with the calculated fraction of seawater to determine empirical Sr distribution coefficients:
-
DSr = (Sr/Ca)solid/(Sr/Ca)fluid.
The PACMANUS end-member hydrothermal fluid calculated by Bach et al. (2003) with data from Douville (1999) has higher Ca (20.1 mM) and Sr (0.114 mM) than seawater (Ca = 10.2 mM; Sr = 0.087 mM). The average calculated empirical Sr distribution coefficient for PACMANUS anhydrites is 0.62 ± 0.21, basically identical to the DSr calculated by Teagle et al. (1998a) for anhydrites from the TAG hydrothermal field (0.62 ± 0.23) and a value of 0.67 provided by Kuhn et al. (2003) for anhydrite from the sediment-hosted Grimsey hydrothermal field north of Iceland. However, apparent DSr values seem to be greater for anhydrite from Site 1188 than for Site 1189 (Fig. F2). The average DSr for Site 1188 is 0.73 ± 0.18, whereas that for Site 1189 is 0.46 ± 0.12.
Chondrite-normalized REE diagrams (Fig. F3A, F3B) depict further discrepancies between anhydrites from Sites 1188 and 1189. Although anhydrites show uniform enrichment of light REE (La to Sm) at both sites, those with peculiar mid-REE (Sm to Dy) enrichments are limited to Site 1188. The overall variability in REE pattern shape and the range in total REE concentration appears greater among the samples from Site 1188. Bach et al. (2003) noted that the REE variability in anhydrite from PACMANUS is much greater than in anhydrites from TAG (Humphris, 1998) and suggested that this difference may indicate a more variable ligand chemistry in the PACMANUS fluids.
REE pattern shape and Sr isotope composition do not appear to be correlated in anhydrites (Fig. F4), indicating that the variations in the proportion of seawater in the fluid does not have a systematic impact on REE contents of anhydrite. In a diagram of 87Sr/86Sr vs. 1000/Sr (Fig. F4A), fluid mixing produces linear trends. Overall, the PACMANUS anhydrite data do not form a systematic trend in Figure F4A, indicating that processes other than fluid mixing have played a role.

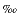 (see discussions in Roberts et al., 2003, and Bach et al., 2003). At seawater salinity, anhydrite precipitates above 140°–150°C when seawater is being heated or seawater (28 mmol/kg sulfate) mixes with nominally sulfate-free hydrothermal fluids. Sr concentrations in the PACMANUS black smoker fluids are slightly higher than those in seawater (Douville, 1999). A simple Sr mass balance provides the proportions of seawater in the fluids from which anhydrite was precipitated:
(see discussions in Roberts et al., 2003, and Bach et al., 2003). At seawater salinity, anhydrite precipitates above 140°–150°C when seawater is being heated or seawater (28 mmol/kg sulfate) mixes with nominally sulfate-free hydrothermal fluids. Sr concentrations in the PACMANUS black smoker fluids are slightly higher than those in seawater (Douville, 1999). A simple Sr mass balance provides the proportions of seawater in the fluids from which anhydrite was precipitated:
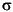 ; n = 105). By comparison, anhydrites recovered from drill core from the Trans-Atlantic Geotraverse (TAG) hydrothermal system at the Mid-Atlantic Ridge 26°N (Mills et al., 1998; Teagle et al., 1998a) precipitated from fluids with an average of 68% ± 12% seawater (1
; n = 105). By comparison, anhydrites recovered from drill core from the Trans-Atlantic Geotraverse (TAG) hydrothermal system at the Mid-Atlantic Ridge 26°N (Mills et al., 1998; Teagle et al., 1998a) precipitated from fluids with an average of 68% ± 12% seawater (1 34S = 21
34S = 21