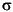 ) of Sr isotope analyses is 30 ppm.
) of Sr isotope analyses is 30 ppm.
Anhydrite samples were separated and analyzed in three different laboratories. At the Woods Hole Oceanographic Institution (WHOI), Massachusetts (USA), Sr isotope and trace element (Sr, Li, Rb, Cs, Ba, La, Ce, Pr, Nd, Sm, Eu, Gd, Tb, Dy, Ho, Er, Yb, Lu, Y, Pb, U, P, and As) analyses were performed. At Southampton Oceanography Center (SOC; UK), Sr and S isotope as well as trace element analyses (Mg, Sr, Rb, Cs, Ba, La, Ce, Pr, Nd, Sm, Eu, Gd, Tb, Dy, Ho, Er, Tm, Yb, Lu, Y, Pb, and U) analyses were carried out. Additional Sr and S isotope analyses were conducted at the Commonwealth Scientific and Industrial Research Organization (CSIRO), New South Wales (Australia).
Anhydrite crystals were carefully hand-picked under a binocular microscope. Several milligrams of anhydrite were gently crushed in a Savillex Teflon beaker and dissolved in dilute HCl at 60°C overnight.
Following sample dissolution, most of the HCl was removed by two cycles of heating to incipient dryness and addition of 1-N HNO3. Sample solutions were centrifuged and injected (via a nebulizer) into a Finnigan element inductively coupled plasma–mass spectrometer (ICP-MS) at WHOI that was calibrated by running matrix-matched trace element standard solutions. Tm and In spikes were added as internal standards. Isobaric mass interferences were checked (e.g., BaO and BaOH interferences on Eu), but oxide production was found negligible, due to the use of a nebulizer. Blanks were monitored and a blank correction was applied. Reproducibility of the data is better than 3%–8%.
About 10 mg of hand-picked anhydrite crystals were dissolved with 2.5-N HCl in Savillex Teflon beakers. A Sr spike was added prior to dissolution to determine Sr concentrations by isotope dilution. Strontium was separated on quartz columns with a 5-mL resin bed of AG50W-X8 200-400 mesh. Sr isotopes were analyzed at WHOI with a VG 354 thermal ionization mass spectrometer (TIMS). 87Sr/86Sr ratios are reported relative to National Bureau of Standards (NBS) reference material 987 = 0.71024. External precision (2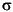 ) of Sr isotope analyses is 30 ppm.
) of Sr isotope analyses is 30 ppm.
Approximately 10–15 mg of anhydrite was weighed into clean Savillex Teflon vials. A 6-mL aliquot of ~2% subboiled nitric acid was added and the samples left to dissolve on a hot plate at 60°C for 10 days.
Following dissolution, the samples were subsampled for Sr and Mg analysis on a Perkin-Elmer Optima 4300 ICP–atomic emission spectrometer (AES) calibrated using synthetic matrix-matched standards. Rare earth elements (REE) and other trace elements were analyzed on a separate subsample using ICP-MS. Calibration was carried out using a suite of matrix-matched synthetic chondritic pattern standards for REE and international rock standards for trace elements.
Subsamples for strontium isotopes were taken to give ~1 µg of Sr; the Sr was separated using 80l Sr-Spec columns eluted with 3-N nitric acid. The samples were loaded with a Ta activator solution onto single Ta filaments and analyzed on a VG Sector 54 TIMS. The value for NBS-987 during this work was 0.710252 ± 18 on 29 runs.
Anhydrite was prepared by microdrilling. Approximately 5–10 mg was required for isotopic analysis. Minor contamination by non S-bearing phases was tolerated and has no effect on the final data. Anhydrite analyses were performed at Scottish Universities Research and Reactor Centre (SURRC; UK) by the technique of Coleman and Moore (1978), in which SO2 gas is liberated by combustion with excess Cu2O and silica at 1125°C. Liberated gases were analyzed on a VG Isotech SIRA II mass spectrometer, and standard corrections applied to raw  66SO2 values to produce true
66SO2 values to produce true  34S. The standards employed were the international standards NBS-123 and IAEA-S-3 and the SURRC standard CP-1. These gave
34S. The standards employed were the international standards NBS-123 and IAEA-S-3 and the SURRC standard CP-1. These gave  34S values of +17.1
34S values of +17.1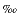 , –31
, –31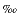 , and –4.6
, and –4.6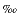 , respectively, with 1
, respectively, with 1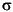 reproducibility better than ±0.2
reproducibility better than ±0.2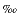 . Data are reported in
. Data are reported in  34S notation as permil (
34S notation as permil (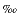 ) variations from the Vienna Canyon Diablo troilite (V-CDT) standard.
) variations from the Vienna Canyon Diablo troilite (V-CDT) standard.
Anhydrite concentrates from selected portions of core were prepared by hand picking under a binocular microscope. After the initial pick, the concentrate was gently ground under acetone and dust removed by elutriation, following which any remaining contaminants (mostly pyrite inclusions) were again hand-picked under the binocular microscope. Where necessary, this was repeated a third time. In this way, concentrates exceeding 99.5% purity and often exceeding 99.9% were obtained. For a few samples with limited quantity, a separate subsample with 1%–2% pyrite contaminant was tolerated for Sr isotope measurements. Bluish gypsum as well as anhydrite was separately picked from Sample 193-1189B-1R-1 (Piece 1A, 0–6 cm); remaining gypsum was removed from the anhydrite concentrate by leaching in boiling water. The concentrate from Sample 193-1189B-3R-1 (Piece 1, 0–11 cm) consisted of 90% gypsum and 10% anhydrite and was submitted in bulk for isotope analysis. The anhydrite concentrate of the thin vein from Sample 193-1188A-17R-1 (Piece 24, 137–140 cm) contained ~10% gypsum and was also submitted in bulk.
For sample dissolution, ~0.01–0.09 g of anhydrite separate was weighed and placed in HCl. The supernatant fluid was decanted ready for cation exchange chromatography. Approximately 0.1g of gypsum was weighed and placed in Milli-Q water to obtain a leachate. The supernatant fluid was decanted, evaporated, and then converted to a chloride form and brought up to final volume in HCl for cation exchange chromatography.
Sr was separated on AG50W–X8 cation exchange resin. The separated Sr was loaded onto a single Ta filament with water and H3PO4 and oxidized in air. The isotopic compositions were measured on a VG 354 TIMS fitted with seven collectors. Samples were run in "dynamic" mode with an ion beam intensity of 3 x 10–11A of 88Sr. Six blocks of nine 87Sr/86Sr ratios were measured, yielding a total of 54 determinations. 87Sr/86Sr ratios were normalized to 86Sr/88Sr = 0.1194 using an exponential correction law. Rubidium was monitored continuously throughout the run, and a negligible correction was applied. The raw data were filtered using a 2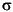 rejection criterion. Measured blanks have a negligible effect on the measured ratios.
rejection criterion. Measured blanks have a negligible effect on the measured ratios.
The precision of individual analyses at 95% confidence limits, determined as two standard errors of the mean, is typically between 0.0010% and 0.0020% (internal precision). Measurements of the standard reference material NBS-987 were made with each batch of samples, yielding an average 87Sr/86Sr ratio of 0.710269 ± 18 (1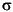 ; n = 33).
; n = 33).
Sulfate was extracted from the anhydrite concentrates by leaching with distilled and degassed water. One gypsum concentrate was treated briefly to avoid dissolution of anhydrite contaminant. The sulfate solutions were separated from undissolved anhydrite, acidified, and BaSO4 was precipitated. The dried BaSO4 precipitate was loaded into a 5-cm- long, 6-mm-diameter quartz tube and dropped into a 9-mm-diameter quartz tube, the system was evacuated, and the sample was heated to >1500°C using a hydrogen/oxygen torch (Bailey and Smith, 1972). The gaseous products were passed over a hot copper plug (600°C) and separated cryogenically. The volume of SO2 was measured and transferred to a suitable vessel for mass spectrometry. Prepared gases were analyzed on a Finnigan 252 mass spectrometer in dual inlet mode. Results are reported using the delta notation relative to V-CDT. Replicate analysis of sulfide and sulfate standard materials were better than ±0.2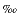 .
.